My experience and observation is when you ferment under pressure there is no "blow off" as you put it. I use 7.5 psi as it is sort of equivalent to a commercial fermenting vessel of approx 5 metres high.Sorry if this has been asked before, but how long can you safely wait after pitching to hook up the serving keg and still feel confident that you have enough co2 to purge it? Really I just want to be wait until there’s no blowoff.
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Keg purging with active fermentation
- Thread starter Mer-man
- Start date

Help Support Homebrew Talk - Beer, Wine, Mead, & Cider Brewing Discussion Forum:
This site may earn a commission from merchant affiliate
links, including eBay, Amazon, and others.
DuncB
Well-Known Member
An oxygen meter checking the liquid that the blow off goes through would indicate when the oxygen in the headspace and beer/ wort has been used up or blown off. Once this phase has been reached which is long before fermentation has finished there will be plenty of CO2 produced to build pressure in a closed system and force starsan out of one or two kegs in my experience.
You have to be a bit more vigilant if you are fast fermenting with Kveik though otherwise you'll miss the ferment.
You have to be a bit more vigilant if you are fast fermenting with Kveik though otherwise you'll miss the ferment.
I would really like to assemble a simple train of items from FV consisting of krausen catcher, CO2resevoir (for cold crash), sanitized corney being purged. I haven't yet succeeded getting all three working well together. The primary problem is krausen getting past the catcher and leaks. The last attempt resulted in the corney not being fully purged, I check with a barBQ lighter and it wasn't extinguished at the opening of the lid. I feel confident that if the system was tight the keg at the end would be properly purged.
I ferment in a 6.5g carboy and fill just below 5.5g mark (OG is typically 060-065 and FG between 009 and 018). I also use fermcap 2 but this doesn't always keep the head down, works well somtimes but not consistant results I use 15 drops minimum.
To eliminate leaks I will switch from rubber bung to carboy cap, my recent krausen barrier/air lock was a 3 ft vertical loop of 1/2 silicone tubing with a few oz of vodka at the bottom of loop, the though was if fermcap let any krausen escape it would be minimal and would mix with the vodka and be contained in the loop, this connected to the CO2 resevoir which was a 3 gal bucket with screw on lid (possible leak source). Lid has two 1/2" barbs installed, the second barb has a hose with black corney QD . I capture the rim of a small unopened kitchen garbage bag around the edge of the bucket as I screw the lid on. The fermentation gas should open the bag fully (corney side not on keg) I have holes drilled in the sides to allow and obverve when the bag is expanded. At that point I would connect the black QD to the keg and vent through a grey QD into starsan container.
When fermentation is finished I would disconnect the QDs from the keg and the CO2 reservoir is waiting for the cold crash cycles. This may all sound crazy but it made sense to me until it didn't work. I have attached a sketch which probably is simpler to understand. I'm hoping someone has incorporated these necessary steps into one setup.
Thanks
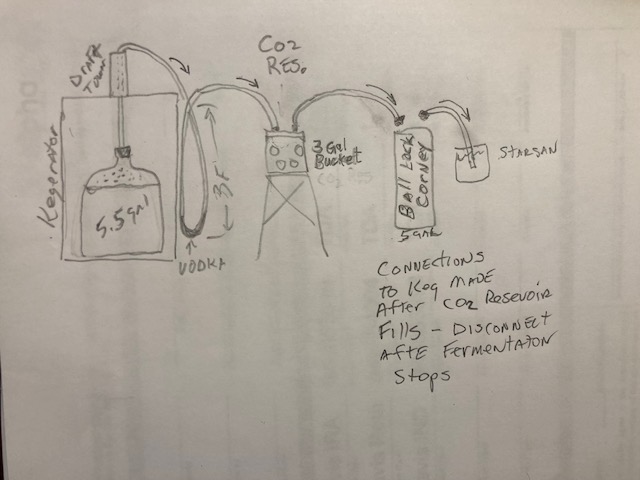
I ferment in a 6.5g carboy and fill just below 5.5g mark (OG is typically 060-065 and FG between 009 and 018). I also use fermcap 2 but this doesn't always keep the head down, works well somtimes but not consistant results I use 15 drops minimum.
To eliminate leaks I will switch from rubber bung to carboy cap, my recent krausen barrier/air lock was a 3 ft vertical loop of 1/2 silicone tubing with a few oz of vodka at the bottom of loop, the though was if fermcap let any krausen escape it would be minimal and would mix with the vodka and be contained in the loop, this connected to the CO2 resevoir which was a 3 gal bucket with screw on lid (possible leak source). Lid has two 1/2" barbs installed, the second barb has a hose with black corney QD . I capture the rim of a small unopened kitchen garbage bag around the edge of the bucket as I screw the lid on. The fermentation gas should open the bag fully (corney side not on keg) I have holes drilled in the sides to allow and obverve when the bag is expanded. At that point I would connect the black QD to the keg and vent through a grey QD into starsan container.
When fermentation is finished I would disconnect the QDs from the keg and the CO2 reservoir is waiting for the cold crash cycles. This may all sound crazy but it made sense to me until it didn't work. I have attached a sketch which probably is simpler to understand. I'm hoping someone has incorporated these necessary steps into one setup.
Thanks

I've done something similar to what you're attempting using this attached to a large Mason jar in the middle of the loop, which should act to filter any particles that get pushed out with the CO2. As you found out it all needs to be sealed air-tight to work. I used hose clamps for this. My setup was much simpler then what you're proposing though, just 1/2" ID tubing coming off my fermenter blowoff, into and out of a Mason jar with krausen catcher (jar half filled with water/StarSan with tubing submerged into the StarSan solution), then over to the liquid-out post on a keg. Had to step it down to fit the QD. Then I attached tubing to the gas-in QD over to a small container of water/StarSan to close the "train." After a few batches I noticed I never had anything ending up in the Mason jar filter so I've since eliminated that piece for simplicity sake, and just connect my blow-off directly to my keg. Seems to work fine for me. My 1/2" tubing coming off my fermenter is about 5 or 6 ft long and I keep it looped. The first foot or so sometimes has a bit of krausen deposited inside, but it never seems to make it all the way to the end of the tubing at the keg.I would really like to assemble a simple train of items from FV consisting of krausen catcher, CO2resevoir (for cold crash), sanitized corney being purged. I haven't yet succeeded getting all three working well together. The primary problem is krausen getting past the catcher and leaks. The last attempt resulted in the corney not being fully purged, I check with a barBQ lighter and it wasn't extinguished at the opening of the lid. I feel confident that if the system was tight the keg at the end would be properly purged.
I ferment in a 6.5g carboy and fill just below 5.5g mark (OG is typically 060-065 and FG between 009 and 018). I also use fermcap 2 but this doesn't always keep the head down, works well somtimes but not consistant results I use 15 drops minimum.
To eliminate leaks I will switch from rubber bung to carboy cap, my recent krausen barrier/air lock was a 3 ft vertical loop of 1/2 silicone tubing with a few oz of vodka at the bottom of loop, the though was if fermcap let any krausen escape it would be minimal and would mix with the vodka and be contained in the loop, this connected to the CO2 resevoir which was a 3 gal bucket with screw on lid (possible leak source). Lid has two 1/2" barbs installed, the second barb has a hose with black corney QD . I capture the rim of a small unopened kitchen garbage bag around the edge of the bucket as I screw the lid on. The fermentation gas should open the bag fully (corney side not on keg) I have holes drilled in the sides to allow and obverve when the bag is expanded. At that point I would connect the black QD to the keg and vent through a grey QD into starsan container.
When fermentation is finished I would disconnect the QDs from the keg and the CO2 reservoir is waiting for the cold crash cycles. This may all sound crazy but it made sense to me until it didn't work. I have attached a sketch which probably is simpler to understand. I'm hoping someone has incorporated these necessary steps into one setup.
Thanks
View attachment 785026
Go to pressure fermenting. I have found 7.5 PSI works well with no carry over to keg being purgedI would really like to assemble a simple train of items from FV consisting of krausen catcher, CO2resevoir (for cold crash), sanitized corney being purged. I haven't yet succeeded getting all three working well together. The primary problem is krausen getting past the catcher and leaks. The last attempt resulted in the corney not being fully purged, I check with a barBQ lighter and it wasn't extinguished at the opening of the lid. I feel confident that if the system was tight the keg at the end would be properly purged.
I ferment in a 6.5g carboy and fill just below 5.5g mark (OG is typically 060-065 and FG between 009 and 018). I also use fermcap 2 but this doesn't always keep the head down, works well somtimes but not consistant results I use 15 drops minimum.
To eliminate leaks I will switch from rubber bung to carboy cap, my recent krausen barrier/air lock was a 3 ft vertical loop of 1/2 silicone tubing with a few oz of vodka at the bottom of loop, the though was if fermcap let any krausen escape it would be minimal and would mix with the vodka and be contained in the loop, this connected to the CO2 resevoir which was a 3 gal bucket with screw on lid (possible leak source). Lid has two 1/2" barbs installed, the second barb has a hose with black corney QD . I capture the rim of a small unopened kitchen garbage bag around the edge of the bucket as I screw the lid on. The fermentation gas should open the bag fully (corney side not on keg) I have holes drilled in the sides to allow and obverve when the bag is expanded. At that point I would connect the black QD to the keg and vent through a grey QD into starsan container.
When fermentation is finished I would disconnect the QDs from the keg and the CO2 reservoir is waiting for the cold crash cycles. This may all sound crazy but it made sense to me until it didn't work. I have attached a sketch which probably is simpler to understand. I'm hoping someone has incorporated these necessary steps into one setup.
Thanks
View attachment 785026
Thanks but how do you compensate when cold crashing? I probably will assemble a krausen catcher for my next batch and maybe use a mylar balloon for the co2 resevoir. Of course a larger FV would solve my blow-off mess probably a 7gal plastic Fermonster will solve the problem.I've done something similar to what you're attempting using this attached to a large Mason jar in the middle of the loop, which should act to filter any particles that get pushed out with the CO2. As you found out it all needs to be sealed air-tight to work. I used hose clamps for this. My setup was much simpler then what you're proposing though, just 1/2" ID tubing coming off my fermenter blowoff, into and out of a Mason jar with krausen catcher (jar half filled with water/StarSan with tubing submerged into the StarSan solution), then over to the liquid-out post on a keg. Had to step it down to fit the QD. Then I attached tubing to the gas-in QD over to a small container of water/StarSan to close the "train." After a few batches I noticed I never had anything ending up in the Mason jar filter so I've since eliminated that piece for simplicity sake, and just connect my blow-off directly to my keg. Seems to work fine for me. My 1/2" tubing coming off my fermenter is about 5 or 6 ft long and I keep it looped. The first foot or so sometimes has a bit of krausen deposited inside, but it never seems to make it all the way to the end of the tubing at the keg.
I get that just not the route I want to goGo to pressure fermenting. I have found 7.5 PSI works well with no carry over to keg being purged
Thanks
With my setup it's easy. I have 30L Speidel fermenter and I bought one of these used from a member here on HBT. It has three ports - blow-off, gas-in, and beer-out. So I just hit it with some CO2 when I cold crash, and can easily add more at any time if I need to.Thanks but how do you compensate when cold crashing? I probably will assemble a krausen catcher for my next batch and maybe use a mylar balloon for the co2 resevoir. Of course a larger FV would solve my blow-off mess probably a 7gal plastic Fermonster will solve the problem.
scrap iron
Well-Known Member
I bought a co2 catcher from Norcal brewing. It's a little pricey but it stores co2 for a cold crash and also prevents krausen and sanitizer from sucking back to the FV.
I have it set up to flush a keg with co2 from the gromet on the second jar. After the ferment is done, I disconnect the line to the keg and cold crash. No krausen is able to enter the keg and no sanitizer can be sucked back into the FV. I use the large 2 qt mason jars to store more co2.
https://www.norcalbrewingsolutions.com/store/CO2-Carbon-Dioxide-Harvester-Kit.html
I have it set up to flush a keg with co2 from the gromet on the second jar. After the ferment is done, I disconnect the line to the keg and cold crash. No krausen is able to enter the keg and no sanitizer can be sucked back into the FV. I use the large 2 qt mason jars to store more co2.
https://www.norcalbrewingsolutions.com/store/CO2-Carbon-Dioxide-Harvester-Kit.html
I have watched their videos and considered this method but was concerned with idea of using a rigid vessel for the co2 unless it was of sufficient volume, which 2 qt may be. If when starting fermentation the 1st jar is filled with starsan which is moved to the 2nd jar as the 1st fills with co2 then the 1st jar volume has to be large enough to handle the volume requirement of cold crashing. Prior to trying to harvest co2 I used a plastic bag I would fill with co2 and connect prior to cold crashing and it would always be collapsed at 45f. My estimation of the bag volume was approx. 1 gallon but I never actually measured it. This was my reason for not using your recommendation.I bought a co2 catcher from Norcal brewing. It's a little pricey but it stores co2 for a cold crash and also prevents krausen and sanitizer from sucking back to the FV.
I have it set up to flush a keg with co2 from the gromet on the second jar. After the ferment is done, I disconnect the line to the keg and cold crash. No krausen is able to enter the keg and no sanitizer can be sucked back into the FV. I use the large 2 qt mason jars to store more co2.
https://www.norcalbrewingsolutions.com/store/CO2-Carbon-Dioxide-Harvester-Kit.html
scrap iron
Well-Known Member
The first jar [to fermenter] is started empty and the second [to air lock or keg] is filled like halfway with sanitizer. During active fermentation the air is pushed out of the first jar and fills with co2 while the second jar acts as the air lock. During cold crash the first jar empties the co2 and the sanitizer starts to suck back into the first. After the sanitizer empties the second jar there is still about a quart of co2 left in it. So, I figure about 3 qts worth of co2 total. I hope I explained it well enough to understand.I have watched their videos and considered this method but was concerned with idea of using a rigid vessel for the co2 unless it was of sufficient volume, which 2 qt may be. If when starting fermentation the 1st jar is filled with starsan which is moved to the 2nd jar as the 1st fills with co2 then the 1st jar volume has to be large enough to handle the volume requirement of cold crashing. Prior to trying to harvest co2 I used a plastic bag I would fill with co2 and connect prior to cold crashing and it would always be collapsed at 45f. My estimation of the bag volume was approx. 1 gallon but I never actually measured it. This was my reason for not using your recommendation.
I have used this system for about 3 years and have not had any suck back of sanitizer or noticed any signs of oxidation. I also rigged a line from the air lock grommet to purge a keg.
I would think the first jar would start out with starsan and as it is pushed to the 2nd pure co2 remains although if starting empty after a full fermentation it would be pure. If I am correct, your volume of co2 to use for crashing is the only the 1st jar as anything returning from the 2nd (following the startsan) would have been mixed with outside airThe first jar [to fermenter] is started empty and the second [to air lock or keg] is filled like halfway with sanitizer. During active fermentation the air is pushed out of the first jar and fills with co2 while the second jar acts as the air lock. During cold crash the first jar empties the co2 and the sanitizer starts to suck back into the first. After the sanitizer empties the second jar there is still about a quart of co2 left in it. So, I figure about 3 qts worth of co2 total. I hope I explained it well enough to understand.
I have used this system for about 3 years and have not had any suck back of sanitizer or noticed any signs of oxidation. I also rigged a line from the air lock grommet to purge a keg.
- Joined
- Jun 4, 2022
- Messages
- 19
- Reaction score
- 23
I'm jumping in to show my solution to purging kegs with CO2.
I use a bucket to ferment to which I added two QD to the lid, one for gas and one for a floating dip tube. I use a plastic hammer to bang the lid onto the bucket to get a good seal. I tested this by putting a few pounds of pressure on it and it held it until I opened the bucket a few days later. Then connect the gas post from the bucket to the in post on the keg and then attach a blow off tube from the key gas post to a pitcher of water. I also put a one way check valve on the blow off tube end that’s submerged in the pitcher of water. This is connected up in my fermentation chamber. When the bubbling stops in a week to 10 days I then do a transfer into the keg. By putting a few pounds of pressure on the bucket via the gas post and attaching the out post to the key out post and then the keg gas post to a blow off tube into a bucket of water, it also has a one way check valve.
Of course the keg was cleaned and sanitized. I also dry hop in the keg by adding the hops before hooking up the tubs in the chamber and use a floating dip tube in the keg. The bucket and the keg are never opened until cleaning time.
So this has improved my beer and seems to be working really well.
I use a bucket to ferment to which I added two QD to the lid, one for gas and one for a floating dip tube. I use a plastic hammer to bang the lid onto the bucket to get a good seal. I tested this by putting a few pounds of pressure on it and it held it until I opened the bucket a few days later. Then connect the gas post from the bucket to the in post on the keg and then attach a blow off tube from the key gas post to a pitcher of water. I also put a one way check valve on the blow off tube end that’s submerged in the pitcher of water. This is connected up in my fermentation chamber. When the bubbling stops in a week to 10 days I then do a transfer into the keg. By putting a few pounds of pressure on the bucket via the gas post and attaching the out post to the key out post and then the keg gas post to a blow off tube into a bucket of water, it also has a one way check valve.
Of course the keg was cleaned and sanitized. I also dry hop in the keg by adding the hops before hooking up the tubs in the chamber and use a floating dip tube in the keg. The bucket and the keg are never opened until cleaning time.
So this has improved my beer and seems to be working really well.
Attachments
From your pictures they look to be carbonation caps, meant to be screwed onto soda bottles. I'm curious, how do you fasten/secure those to the lid, so they don't pop out? I can't tell from pix #3 how they're held in place.I use a bucket to ferment to which I added two QD to the lid, one for gas and one for a floating dip tube.
- Joined
- Jun 4, 2022
- Messages
- 19
- Reaction score
- 23
You are correct, they are carbonation caps. I cut the the tops off a couple of plastic bottles and used that on the inside of the lid to attach the caps. Also needed some large rubber washers on the outside, one per cap, to get a good seal. I tried the cheaper plastic carbonation caps but never got a good seal so I tried the metal caps that worked.From your pictures they look to be carbonation caps, meant to be screwed onto soda bottles. I'm curious, how do you fasten/secure those to the lid, so they don't pop out? I can't tell from pix #3 how they're held in place.
I understand your set up but have a couple questions.I'm jumping in to show my solution to purging kegs with CO2.
I use a bucket to ferment to which I added two QD to the lid, one for gas and one for a floating dip tube. I use a plastic hammer to bang the lid onto the bucket to get a good seal. I tested this by putting a few pounds of pressure on it and it held it until I opened the bucket a few days later. Then connect the gas post from the bucket to the in post on the keg and then attach a blow off tube from the key gas post to a pitcher of water. I also put a one way check valve on the blow off tube end that’s submerged in the pitcher of water. This is connected up in my fermentation chamber. When the bubbling stops in a week to 10 days I then do a transfer into the keg. By putting a few pounds of pressure on the bucket via the gas post and attaching the out post to the key out post and then the keg gas post to a blow off tube into a bucket of water, it also has a one way check valve.
Of course the keg was cleaned and sanitized. I also dry hop in the keg by adding the hops before hooking up the tubs in the chamber and use a floating dip tube in the keg. The bucket and the keg are never opened until cleaning time.
So this has improved my beer and seems to be working really well.
Is the FV volume large enough to prevent krausen from getting into keg?
Do you cold crash and if so what is the effect on the closed system, is the keg still connected?
- Joined
- Jun 4, 2022
- Messages
- 19
- Reaction score
- 23
The krausen has not been a problem, I do 5 gallon batches and the bucket is 7.8 gallons, there is enough head room.I understand your set up but have a couple questions.
Is the FV volume large enough to prevent krausen from getting into keg?
Do you cold crash and if so what is the effect on the closed system, is the keg still connected?
After transfering to the keg after about a week I leave at room temp for another week, some times longer, then chill. The keg is more like a secondary fermentation and then cold crashing the keg would not be an issue because it's sealed tight and after a week at room temp there will be some pressure in the keg so you don't have issues like cold crashing in the fermentor.
I trust the math from Doug293cz
https://www.homebrewtalk.com/threads/keg-purging-with-active-fermentation.628658/post-8004741
From your pictures they look to be carbonation caps, meant to be screwed onto soda bottles. I'm curious, how do you fasten/secure those to the lid, so they don't pop out? I can't tell from pix #3 how they're held in place.
Note that for a similar solution, you could also use a "Ball Lock Post Bulkhead". I have a few of these that I added to a lid for my Fermonster. The downside of using ball lock disconnects is that it makes for a very narrow passage that could easily get clogged with krausen or hops. I try to make sure I have plenty of headspace or I am working with a yeast known for less of a krausen. Temp control helps as well.
Bulkhead Posts:
https://www.morebeer.com/products/body-connect-gas-ball-lock-post-bulkhead-assembly.html
- Joined
- Jun 4, 2022
- Messages
- 19
- Reaction score
- 23
Note that for a similar solution, you could also use a "Ball Lock Post Bulkhead". I have a few of these that I added to a lid for my Fermonster. The downside of using ball lock disconnects is that it makes for a very narrow passage that could easily get clogged with krausen or hops. I try to make sure I have plenty of headspace or I am working with a yeast known for less of a krausen. Temp control helps as well.
Bulkhead Posts:
https://www.morebeer.com/products/body-connect-gas-ball-lock-post-bulkhead-assembly.html
Yes, I tried these first but was not able to get a good seal. The weight of the connections would pull on the post and break the seal. The lids are too flexible for the narrow post. The carbonation caps have a much wider base and hold the seal very well after the connections are in place.
Attached is a short video of the current batch in the chamber, it was brewed on Sunday and the fermenation is in high gear. I hope you can hear the bubbling.
Attachments
When enlarging the images, I can see the raw edges of the bottle tops on the inside.I cut the the tops off a couple of plastic bottles and used that on the inside of the lid to attach the caps.
I guess it's the rim on the top of the bottle, underneath the threads, that makes the actual seal with the flat (?) rubber washer that's on the inside.
Then the carb cap sit on another flat washer on the outside? The lid being sandwiched between the 2 flat rubber washers?
- Joined
- Jun 4, 2022
- Messages
- 19
- Reaction score
- 23
Yes, washer on both sides, it was trial and error and I destroyed a couple of lids in the process. Be sure to find a flat and smooth part of the lid to drill the holes, any raised letters or other artifacts will cause issues with the seal. Trust me on this point.When enlarging the images, I can see the raw edges of the bottle tops on the inside.
I guess it's the rim on the top of the bottle, underneath the threads, that makes the actual seal with the flat (?) rubber washer that's on the inside.
Then the carb cap sit on another flat washer on the outside? The lid being sandwiched between the 2 flat rubber washers?
I got the idea from the attached video, he goes into detail on how to prep the bottle part, there is a part near the threads that has to be cut off.
Of course I put the caps on the bucket lid.
Attachments
I trust his math also that was not the issue.The krausen has not been a problem, I do 5 gallon batches and the bucket is 7.8 gallons, there is enough head room.
After transfering to the keg after about a week I leave at room temp for another week, some times longer, then chill. The keg is more like a secondary fermentation and then cold crashing the keg would not be an issue because it's sealed tight and after a week at room temp there will be some pressure in the keg so you don't have issues like cold crashing in the fermentor.
I trust the math from Doug293cz
https://www.homebrewtalk.com/threads/keg-purging-with-active-fermentation.628658/post-8004741
- Joined
- Jun 4, 2022
- Messages
- 19
- Reaction score
- 23
I trust his math also that was not the issue.
I have not had any issues with krausen because there is plenty of head space in the bucket.
The part about the math was unrelated to the krausen question, that wasn't very clear in my response. I was adding that I trust that there is enough CO2 to purge the keg being produced based on what Doug293cz posted.
scrap iron
Well-Known Member
I'm posting a follow up on my post # 171 to let others know how my last few cold crashes went.
Earlier when I cold crashed, I just set the temp on my fermentation fridge to 50 degrees and let er rip. I bought a new fridge less than a year ago and have been concerned with over working the compressor. So, I have been reducing the temp 5 degrees at a time and letting it rest for a while before going lower. These last few times seemed to have lessened the amount of sanitizer being sucked back to the first jar. It just so happens I started a cold crash on a Strong Bitter Tuesday evening and finishing Wednesday evening, about 24 hours' time. I got it down to 50 degrees from 68 and only half the sanitizer was sucked back leaving no air co2 mix to enter the first jar.
I think the gradual reducing of the temperature helped with less suck back. Not scientific I know but I have noticed an improvement.
Any way I thought I'd share my experience.
Earlier when I cold crashed, I just set the temp on my fermentation fridge to 50 degrees and let er rip. I bought a new fridge less than a year ago and have been concerned with over working the compressor. So, I have been reducing the temp 5 degrees at a time and letting it rest for a while before going lower. These last few times seemed to have lessened the amount of sanitizer being sucked back to the first jar. It just so happens I started a cold crash on a Strong Bitter Tuesday evening and finishing Wednesday evening, about 24 hours' time. I got it down to 50 degrees from 68 and only half the sanitizer was sucked back leaving no air co2 mix to enter the first jar.
I think the gradual reducing of the temperature helped with less suck back. Not scientific I know but I have noticed an improvement.
Any way I thought I'd share my experience.
ihavenonickname
Well-Known Member
What’s the concensus on purging kegs with co2 fermentation? Best methods? Do I need to fill the serving keg with sanitizer so it gets pushed out? Can I just sanitize the kegs and leave it empty to get flushed out by the 300+ liters of co2 produces? It’s easier that way.
What’s a reasonable head space to do this with? If Normal head space is 2 gallons, will it effectively purge 7 gallons? ( +serving keg) will it effectively purge 12 gallons? (+a serving keg AND a dry hop keg?)
I’ve been playing around with my method and I’ve had some really lackluster aroma on my IPAs lately as I’ve gone to this purge a serving and dry hop keg. Maybe this process is to blame? Not sure how to do it but always looking for better aroma and a simple process.
My hunch is actually pushing out sanitizer can only help, but it might be overkill.
What’s a reasonable head space to do this with? If Normal head space is 2 gallons, will it effectively purge 7 gallons? ( +serving keg) will it effectively purge 12 gallons? (+a serving keg AND a dry hop keg?)
I’ve been playing around with my method and I’ve had some really lackluster aroma on my IPAs lately as I’ve gone to this purge a serving and dry hop keg. Maybe this process is to blame? Not sure how to do it but always looking for better aroma and a simple process.
My hunch is actually pushing out sanitizer can only help, but it might be overkill.
IMO if you fully sanitized keg after initially cleaning then no need to push out with CO2
Adam Zerwick
Well-Known Member
- Joined
- Apr 13, 2018
- Messages
- 74
- Reaction score
- 35
What’s the concensus on purging kegs with co2 fermentation? Best methods? Do I need to fill the serving keg with sanitizer so it gets pushed out? Can I just sanitize the kegs and leave it empty to get flushed out by the 300+ liters of co2 produces? It’s easier that way.
What’s a reasonable head space to do this with? If Normal head space is 2 gallons, will it effectively purge 7 gallons? ( +serving keg) will it effectively purge 12 gallons? (+a serving keg AND a dry hop keg?)
I’ve been playing around with my method and I’ve had some really lackluster aroma on my IPAs lately as I’ve gone to this purge a serving and dry hop keg. Maybe this process is to blame? Not sure how to do it but always looking for better aroma and a simple process.
My hunch is actually pushing out sanitizer can only help, but it might be overkill.
Based on the maths from @doug293cz you'd be left with the following O2 in the headspace (fermenter and kegs). This is assuming a 7g fermenter with 5g of 1.060 wort in it and an estimated FG of 1.012
7g headspace: 1.4 ppb O2
12g headspace: 3511 ppb O2
My approach when using a separate dry hop keg was:
1) Use blowoff on fermenter until the SG where the headspace in fermenter is calculated to be 1 ppb
2) Attach fermenter to serving keg and push out sanitizer with fermentation CO2
3) Once sanitizer is fully pushed out detach serving keg
4) Attach dry hop keg (loaded with dry hops) to fermenter and flush with fermentation CO2.
5) Optional... Cap the dry hop keg and spund with a few points left.
I use a floating hydrometer to know roughly what the SG is and I have my own spreadsheet to do all these calculations for me.
IMO if you fully sanitized keg after initially cleaning then no need to push out with CO2
The "CO2 Star San Push" at that point isn't actually for sanitation, it's for purging all but CO2 out of a keg before filling it. In the discussion prior it's an alternative to using fermentation gases to purge the keg(s).
Which is what I'm literally doing right now: the fridge is plumbed for fermentation gas (out the upper right corner). The plastic jug serves as a foam catcher and consolidates the two carboy feeds into one line.


That line passes through the bulkhead and runs around to the pair of daisy-chained kegs leading to a water bottle which is merrily bubbling away
And, yes, it works, and is easier than a "Star San Purge", which I used to do until HBT's resident fluid physics guy @doug293cz proved it should work...
Cheers!
ihavenonickname
Well-Known Member
Whoa that’s a huge difference 7g vs 12g! Is there a decimal missing?Based on the maths from @doug293cz you'd be left with the following O2 in the headspace (fermenter and kegs). This is assuming a 7g fermenter with 5g of 1.060 wort in it and an estimated FG of 1.012
7g headspace: 1.4 ppb O2
12g headspace: 3511 ppb O2
My approach when using a separate dry hop keg was:
1) Use blowoff on fermenter until the SG where the headspace in fermenter is calculated to be 1 ppb
2) Attach fermenter to serving keg and push out sanitizer with fermentation CO2
3) Once sanitizer is fully pushed out detach serving keg
4) Attach dry hop keg (loaded with dry hops) to fermenter and flush with fermentation CO2.
5) Optional... Cap the dry hop keg and spund with a few points left.
I use a floating hydrometer to know roughly what the SG is and I have my own spreadsheet to do all these calculations for me.
Yeah so I’ve been trying to think through a procedure like you outlined… it gets a bit complicated! But I don’t think I follow the logic. Why not just start the ferment with the dry hop keg attached and the serving keg next in series? Maybe then fill the serving keg with star San so it gets pushed out? That would effectively reduce the headspace back to 7g, right? Is that part even that helpful tho?
I’d be interested in playing with the spread sheet if you can share.
Adam Zerwick
Well-Known Member
- Joined
- Apr 13, 2018
- Messages
- 74
- Reaction score
- 35
Whoa that’s a huge difference 7g vs 12g! Is there a decimal missing?
Yeah so I’ve been trying to think through a procedure like you outlined… it gets a bit complicated! But I don’t think I follow the logic. Why not just start the ferment with the dry hop keg attached and the serving keg next in series? Maybe then fill the serving keg with star San so it gets pushed out? That would effectively reduce the headspace back to 7g, right? Is that part even that helpful tho?
I’d be interested in playing with the spread sheet if you can share.
Here's how I understand things... The fermentation only produces a certain amount of CO2 which you can use to displace stuff in the headspace. If that headspace is just air then the headspace contains roughly 20% O2 and the CO2 from fermentation ends up diluting the headspace due to how gases mix. It could be that this dilution and the math I've used from @doug293cz is the worst case and the real world is better. In any event this dilution with your 12g headspace ends up with 3511 ppb according to the math.
My approach hinges on the idea that the fermenter headspace contains roughly 20% O2 prior to fermentation starting and doesn't get absorbed into the wort and therefore isn't consumed by the yeast. With that idea in mind (the worst case) the O2 would mix/dilute the rest of the headspace (other kegs) if you were to daisy chain them. So why not hook up a blow off to the fermenter so that only the fermenter's headspace is diluted with fermentation CO2 until the point that the fermenter headspace is entirely CO2? At that point any gas exiting the fermenter is essentially pure CO2 and can be used to purge/flush whatever vessel you want. It's as if the fermenter is a CO2 tank at that point. Is this approach overkill? Maybe but I don't mind.
Keg Purge Final O2
ihavenonickname
Well-Known Member
Thanks! I must be missing a critical concept that the headspace o2 is 3000x worse when the head space is only ~double the size (12g vs 7g). ??
Yeah I like a get your procedure except for how the numbers work out that there is an dvantage to stage out the process... why does your 2g headspace drop to 1ppb fast enough to still fully purge out a DH keg?
appreciate you engaging me on this topic! we are pretty far down the beer math nerd rabbit hole, but I can't stop thinking about it!
Yeah I like a get your procedure except for how the numbers work out that there is an dvantage to stage out the process... why does your 2g headspace drop to 1ppb fast enough to still fully purge out a DH keg?
appreciate you engaging me on this topic! we are pretty far down the beer math nerd rabbit hole, but I can't stop thinking about it!
DuncB
Well-Known Member
There isn't a consensus, this thread has lots of detail.What’s the concensus on purging kegs with co2 fermentation? Best methods?
My hunch is actually pushing out sanitizer can only help, but it might be overkill.
I purge out sanitiser with ferment gas once ferment has been underway for a few days.
I also put some ascorbic acid in the keg closed technique before the closed transfer.
See thread I think called Vitamin C the game changer or ascorbic acid the game changer.
Adam Zerwick
Well-Known Member
- Joined
- Apr 13, 2018
- Messages
- 74
- Reaction score
- 35
Thanks! I must be missing a critical concept that the headspace o2 is 3000x worse when the head space is only ~double the size (12g vs 7g). ??
Yeah I like a get your procedure except for how the numbers work out that there is an dvantage to stage out the process... why does your 2g headspace drop to 1ppb fast enough to still fully purge out a DH keg?
appreciate you engaging me on this topic! we are pretty far down the beer math nerd rabbit hole, but I can't stop thinking about it!
I suggest you read through the brilliant post from @doug293cz to see how each bubble of CO2 produced affects things
Maths
This is the equation that all this centers on:
Final O2 Conc = 210000 ppm * ((Headspace volume L - 0.0001 L) / Headspace volume L)^ (Liters of CO2 produced / .0001 L)
From the assumptions I made, 5g 1.060 wort with FG of 1.012 you'll produce 499.35 L of CO2. Your headspace is either 7g or 12g. Plugging this into the equation:
210000 * ((26.5 L - .0001L) / 26.5 L) ^ (499.35 L/.0001 L) = .00137 ppm
210000 * ((45.4 L - .0001L) / 45.4 L) ^ (499.35 L/.0001 L) = 3.51 ppm
How this O2 in the headspace dissolves into solution is beyond me. Maybe somebody smarter than me can shed some light here. My take is that these calculations are all worst case scenario so real world is likely better than this however given enough time, agitation, etc. at a given temperature the O2 will be absorbed into the beer.
You can use that same equation to figure out how many liters of CO2 is required to hit 1 ppb in 2g of headspace. Turns out it takes 145.6 L of CO2 which is roughly when your SG drops from 1.060 to 1.046. At this point we can assume the fermenter is exhausting pure CO2. To purge/displace the serving keg full of sanitizer takes the volume of serving keg worth of CO2 which is 18.9 L. The SG when this has completed should be about 1.044. From there the remainder of the CO2 produced from 1.044 to FG of 1.012 is used to dilute the O2 in the dry hop keg. In my calculations I've assumed the CO2 filled fermenter does not mix with the gas in the dry hop keg... This will result in the dry hop keg with 4.25 ppb O2.
ihavenonickname
Well-Known Member
This addresses my biggest question... but your response here is not consistent with your equation (If I'm using it right!) When you change the total headspace from 7 gal to 12 gal (one keg to purge vs two kegs) the o2 left in the system increases from ~2ppb to ~4000ppb. I'd really like to think that I can purge two kegs for my dry hop method, can you explain your thinking to me?! I wantOk, a "5" gal corny has an actual volume of about 5.35 gal, so 2 of those plus 1.5 gal headspace in the fermenter totals 12.2 gal, or 46.2 liters. Assuming you have 20 liters of wort at 1.050 that finishes at 1.010, you would generate ~440 liters of CO2 during fermentation. After some multiple gas dilution cycles the final O2 concentration is about 15 ppb (0.015 ppm.) So, purging (2X) 5 gal kegs with a 5 gal ferment appears to give acceptable results.
Brew on
ihavenonickname
Well-Known Member
Thanks yes great explanation. I'm following your math and thinking now. I guess its just very strategic use of the effects of compounding. Maybe I should hire you as my personal finance, retirement planner? ha!I suggest you read through the brilliant post from @doug293cz to see how each bubble of CO2 produced affects things
Maths
This is the equation that all this centers on:
Final O2 Conc = 210000 ppm * ((Headspace volume L - 0.0001 L) / Headspace volume L)^ (Liters of CO2 produced / .0001 L)
From the assumptions I made, 5g 1.060 wort with FG of 1.012 you'll produce 499.35 L of CO2. Your headspace is either 7g or 12g. Plugging this into the equation:
210000 * ((26.5 L - .0001L) / 26.5 L) ^ (499.35 L/.0001 L) = .00137 ppm
210000 * ((45.4 L - .0001L) / 45.4 L) ^ (499.35 L/.0001 L) = 3.51 ppm
How this O2 in the headspace dissolves into solution is beyond me. Maybe somebody smarter than me can shed some light here. My take is that these calculations are all worst case scenario so real world is likely better than this however given enough time, agitation, etc. at a given temperature the O2 will be absorbed into the beer.
You can use that same equation to figure out how many liters of CO2 is required to hit 1 ppb in 2g of headspace. Turns out it takes 145.6 L of CO2 which is roughly when your SG drops from 1.060 to 1.046. At this point we can assume the fermenter is exhausting pure CO2. To purge/displace the serving keg full of sanitizer takes the volume of serving keg worth of CO2 which is 18.9 L. The SG when this has completed should be about 1.044. From there the remainder of the CO2 produced from 1.044 to FG of 1.012 is used to dilute the O2 in the dry hop keg. In my calculations I've assumed the CO2 filled fermenter does not mix with the gas in the dry hop keg... This will result in the dry hop keg with 4.25 ppb O2.
still curious what @doug293cz thinks about my comment above.
Yes, exactly. The calculations are indeed "worst case", as has been stated several times in this thread IIRC. The assumption that evolved CO2 and headspace gases completely homogenize before any of the headspace gas gets displaced out the final exhaust is extremely pessimistic. But this assumption was necessary to be able to do any calculations to estimate the effectiveness of ferm gas purging. I have been told (by someone I believe) that measurements have been made, with equipment sensitive enough to measure ppb levels of O2, showing that residual O2 levels are significantly lower than the calculations predict. This is actually a good thing, as we know the process is extremely robust.How this O2 in the headspace dissolves into solution is beyond me. Maybe somebody smarter than me can shed some light here. My take is that these calculations are all worst case scenario so real world is likely better than this however given enough time, agitation, etc. at a given temperature the O2 will be absorbed into the beer.
Brew on

This addresses my biggest question... but your response here is not consistent with your equation (If I'm using it right!) When you change the total headspace from 7 gal to 12 gal (one keg to purge vs two kegs) the o2 left in the system increases from ~2ppb to ~4000ppb. I'd really like to think that I can purge two kegs for my dry hop method, can you explain your thinking to me?! I want
I haven't messed around with the math for this since I wrote the original analysis. I'm going to go back and check some of the other implementations of my equations, as I did a similar 1 keg vs. 2 keg purge comparison some time ago, and got very different results.Thanks yes great explanation. I'm following your math and thinking now. I guess its just very strategic use of the effects of compounding. Maybe I should hire you as my personal finance, retirement planner? ha!
still curious what @doug293cz thinks about my comment above.
Brew on

Bark0s
Member
Here's a link to the threadThere isn't a consensus, this thread has lots of detail.
I purge out sanitiser with ferment gas once ferment has been underway for a few days.
I also put some ascorbic acid in the keg closed technique before the closed transfer.
See thread I think called Vitamin C the game changer or ascorbic acid the game changer.
https://www.homebrewtalk.com/threads/vitamin-c-the-game-changer.698328/
DuncB
Well-Known Member
Yes that's the one. Thank you. Thread a little dormant of late.
kurds_2408
Well-Known Member
- Joined
- Dec 19, 2013
- Messages
- 218
- Reaction score
- 167
I’m plannig to copy this exact setup. Found this thread after having an idea for something similar but not sure how to start. All the other options are to complicated for my simple homebrewing and only own buckets currently. Just designed this little thing up for my 3D printer since I don’t have any coke bottles. Should give more support than a bottle top and easier to tighten too.I'm jumping in to show my solution to purging kegs with CO2.
I use a bucket to ferment to which I added two QD to the lid, one for gas and one for a floating dip tube. I use a plastic hammer to bang the lid onto the bucket to get a good seal. I tested this by putting a few pounds of pressure on it and it held it until I opened the bucket a few days later. Then connect the gas post from the bucket to the in post on the keg and then attach a blow off tube from the key gas post to a pitcher of water. I also put a one way check valve on the blow off tube end that’s submerged in the pitcher of water. This is connected up in my fermentation chamber. When the bubbling stops in a week to 10 days I then do a transfer into the keg. By putting a few pounds of pressure on the bucket via the gas post and attaching the out post to the key out post and then the keg gas post to a blow off tube into a bucket of water, it also has a one way check valve.
Of course the keg was cleaned and sanitized. I also dry hop in the keg by adding the hops before hooking up the tubs in the chamber and use a floating dip tube in the keg. The bucket and the keg are never opened until cleaning time.
So this has improved my beer and seems to be working really well.

Similar threads
- Replies
- 2
- Views
- 521
- Replies
- 14
- Views
- 1K
- Replies
- 5
- Views
- 322







